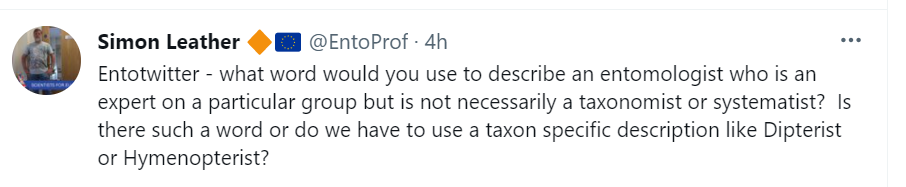
Insects really took off when they developed flight (Alexander, 2015), so it is perhaps surprising that so many have lost the ability subsequently. Nearly all the winged Orders have developed flightless members, with beetles of course, topping the list (Wagner & Liebherr, 1992). A number of reasons for why flightlessness made a reappearance have been put forward. The eminent coloepterist, Thomas Vernon Wollaston, noted that the island of Madeira had an unusually high number of wingless (apterous) beetles. His friend, Charles Darwin, suggested that for island dwelling animals, it was a disadvantage to be winged especially if you were small or subjected to high winds (Darwin, 1859). Many years later, Derek Roff reviewed the literature, and found that there was no difference in the proportion of non-winged insects on islands compared with those on continental areas (Roff, 1990). Winglessness is also common in insects living at high altitudes, in cold climates or in those that are autumn or winter active (Hackman, 1966). It might be that wings are energetically costly in those environments (Mani, 1962), but why then is it that in many cases, it is only the females that are wingless? To explain this we can hypothesise that eggs are energetically more expensive than sperm (Hayward & Gilooly, 2011), so that males can ‘afford’ to be winged and travel to find a mate. For this to work, the females need to be able to attract males from a distance, something moths are renowned for (Greenfield, 1981).

Male Thyridopteryx ephemeraeformis – note the well-developed antenna – ideal for picking up female sex pheromones. https://upload.wikimedia.org/wikipedia/commons/4/4d/-_0457_%E2%80%93_Thyridopteryx_ephemeraeformis_%E2%80%93_Evergreen_Bagworm_Moth_%2814869905567%29.jpg By Andy Reago & Chrissy McClarren [CC BY 2.0 (http://creativecommons.org/licenses/by/2.0)%5D, via Wikimedia Commons
We also know that in those insects with wing dimorphism, the apterous forms are more fecund compared with those with wings (Dixon, 1972; Mackay & Wellington, 1975). In those insects that retain their wings, many resorb their wing muscles once they have found suitable egg laying sites (Stjernholm et al., 2005; Tan et al., 2010), further proof that wings are costly. Winglessness is also common in those insects that are parasitic on vertebrates, bedbugs, fleas and lice for example. Those that do start with wings, such as the Hippoboscid flies, lose their wings once they have found a suitable host. Finally, winglessness is often associated with stable and extensive habitats, such as forests, or surprisingly to me at any rate, mountains, where dispersal is not a high priority (Roff, 1990).

Thyridopteryx ephemaeraeformis https://ideastations.org/sites/default/files/storage/secondary-images/bagworm-case.jpg
I first saw bagworms as a child in Jamaica but of course at the time had no idea what species they were. I was however, fascinated by the sight of the cunningly constructed cases in which the larvae lived and eventually pupated within. To me, case bearer moths and caddisflies were the insect equivalent of hermit crabs, which were and are one of my favourite non-insect animals*. Little did I know that one day I would write about these very same bagworms (Rhainds et al., 2008). Bagworms, of which just over half have wingless females, immediately contradict the cold climate hypothesis of winglessness, as many of them are tropical and there are just as many wingless species in the tropics as there are elsewhere (Rhainds et al., 2009). The bagworms belong to the family Pyschidae, which contain a 1000 species or so. Not only do over half of these have wingless females, some also have females which are legless and never leave their pupal case, even mating in it.

Male Thyridopteryx ephemeraeformis bagworm mating with bag-enclosed female (Jones, 1927)
Even though the more primitive (less derived) members of the Psychidae have wings, the winged females are less active than the males (Rhainds et al. 2009). As you might expect, host plant selection is by the larval stage, which on hatching, throw out a silk thread and float off with great expectations (Moore & Hanks, 2004). Once they find a suitable host plant, which is not as difficult as you might expect, as they extremely polygamous, they begin to feed and construct their cases. Some of the larval cases that Psychids construct are truly magnificent. A great example is Eumeta crameri, the large faggot worm, so called because it looks like it is carrying a pile of firewood on its back 🙂

Eumeta crameri, the large faggot worm, so called because of the twigs its carries around on its back Melvyn Yeo
In case you are wondering about the ornate cases, they are not decorative, but more likely to be anti-predator devices (Khan, 2020).
Although the Psychids have the largest number of species with wingless females, there are 18 other moth families with species with wingless females. Species that are found at high altitudes and northern latitudes have the most flightless species (Hackmann, 1966) or, like the Psychids, inhabit stable forest and woodland habitats (Barbosa et al., 1989). Another characteristic of wingless moth species is that they overwinter as eggs or first instar larvae (Barbosa et al., 1989, although there are of course, many moths that have similar habits and are not wingless, such as the small ermine moths (Leather, 1986a).
After the Psychids, the families with the greatest number of species with wingless females are the Geometridae (loopers) and the Lymantridae (tussock moths). In the Lymantridae some are wingless and many have non-functional wings (Hackman, 1966). The Arctidae and the Lasiocampidae also have some flightless species, the genus Chondrostega, endemic to the Iberian Peninsula having some notable examples, (Hackman, 1966). An oddity, as they are not strictly flightless, are females of the tortricid Choristoneura fumiferana, which have functional wings, but are behaviourally flightless, only taking flight under particular environmental conditions (Barbosa et al., 1989).
Moth species that have flightless females all have one thing in common, they aren’t picky about their diet, they are polyphagous and live in forests and woodlands. They also tend to have larvae that can disperse by ballooning, although not all moths with ballooning larvae have flightless females. First instar larvae of the pine beauty moth, Panolis flammea, which readily balloon in outbreak situations, and usefully, can survive several days without food (Leather, 1986b).
In the UK there are two very common moths with wingless females, the winter moth, Operphthera brumata and the Vapourer moth, Orgyia antiqua, the former a Geoemtrid, the latter, a Lymantrid. Both are extremely polyphagous, usually feeding on broadleaf trees and shrubs, but both have recently added conifer species to their diets. The Vapourer moth ‘decided’ that the introduced lodgepole pine, Pinus contorta growing in Sutherland and Caithness, would make a suitable alternative food plant (Leather, 1986) and the winter moth opted for another introduced conifer, Sitka spruce, Picea sitchensis, in the Scottish Borders (Hunter et al., 1991). Why both these host shifts happened in the early 1980s and in Scotland, remains a mystery, although it is possible that they moved onto conifers via heather (Hewson & Mardon, 1970; Kerslake et al., 1996).
They do, however, have some striking differences in their approach to life. Larvae of the Winter moth are spring flush feeders, and very dependent on egg hatch coinciding with bud burst (Wint, 1983), Vapourers are summer foliage feeders so are adapted to feeding on mature leaves. The adults of the Winter moth, as its name suggests are active in the winter months, laying their eggs on the bark or in crevices of their host trees in November and December and even January. Vapourer adults on the other hand are summer active, the eggs being laid on their pupal cases on the leaves of their host trees from July to September.

Female Vapourer moth and her egg mass – note the short legs and much reduced wings

Long legged female winter moth Operphtera brumata https://butterfly-conservation.org/sites/default/files/styles/large/public/2019-01/8179909985_76865dd047_o.jpg
Hackman (1966) distinguishes two types of wingless females, those with reduced locomotion, very heavy, filled with eggs and what I describe in class as splurgers, i.e. all their eggs laid in one go. The female Vapourer with short legs and much-reduced wings is an ideal example. The female winter moth is a good example of the second type, those possessing good strong legs which after copulation seek out suitable egg-laying sites. Despite the difference in oviposition tactics, the first instar larvae of both species are adept ballooners, and it is they who ‘decide’ whether to stay or go (Tikkanen et al., 1999).

First instar Vapourer moth larvae in the process of dispersing.
Understandably, they have very little control of where they land, although presumably, they can reject the plant they land on and launch themselves into space again. How many times they can do this and how long they can live for without feeding, is something that needs research, but given that the first instar larvae of the pine specialist P. flammea can live several days without feeding, I would expect that the Winter moth and Vapourer moth larvae are equally capable of resisting starvation.
Moths without wings, but highly successful and many are pests, so not such a dumb approach to life after all?

And while we’re at it, here is the lymantriid Teia anartoides. With hamsterlike apterous females! AinsleyS @americanbeetles
References
Alexander, D.E. (2015) On the Wing, Oxford University Press. (This is an excellent book).
Barbosa, P., Krischik, V. & Lance, D. (1989) Life history traits of forest-inhabiting flightless lepidoptera. American Midland Naturalist, 122, 262-274.
Darwin, C. (1859) 0n the Origin of Species, JohnMurray, \lodnon.
Dixon, A.F.G. (1972) Fecundity of brachypterus and macropterous alatae in Drepanosiphum dixoni (Callaphididae, Aphididae). Entomologia experimentalis et applicata, 15, 335-340.
Greenfield, M.D. (1981) Moth sex pheromones: an evolutionary perspective. Florida Entomologist, 64, 4-17.
Hackman, W. (1966) On wing reduction and loss of wings in Lepidoptera. Notulae Entomologicae, 46, 1-16.
Hayward, A. & Gillooly, J.F. (2011) The cost of sex: quantifying energetic investment in gamete production by males and females. PLoS One, 6, e16557
Hewson, R. & Mardon, D.K. (1970) Damage to heather moorland by caterpillars of the vapourer moth Orgyia antiqua L. (Lep., Lymantridae). Entomologist’s Monthly Magazine, 106, 82-84.
Hunter, M.D., Watt, A.D. & Docherty, M. (1991) Outbreaks of the winter moth on Sitka spruce are not influenced by nutrient deficiencies of trees. Oecologia, 86, 62-69.
Jones, F.M. (1927) Mating of the Psychidae (Lepidoptera). Transactions of the Entomological Society of America, 53, 293-312.
Kerslake, J.E., Kruuk, L.E.B., Hartley, S.E. & Woodin, S.J. (1996) Winter moth (Operophtera brumata (Lepidoptera: Geometridae)) outbreaks on Scottish moorlands; effects of host plant and parasitoids on larval survival and development. Bulletin of Entomological Research, 86, 155-164.
Khan, M.K. (2020) Bagworm decorations are an anti-predator structure. Ecological Entomology https://onlinelibrary.wiley.com/doi/epdf/10.1111/een.12876
Leather, S.R. (1986a) Insects on bird cherry I The bird cherry ermine moth, Yponomeuta evonymellus (L.). Entomologist’s Gazette, 37, 209-213.
Leather, S.R. (1986b) The effect of neonatal starvation on the growth, development and survival of larvae of the pine beauty moth Panolis flammea. Oecologia, 71, 90-93.
Leather, S.R. (1986c) Keep an eye out for the vapourer moth. Forestry & British Timber, 15, 13.
Mackay, P.A. & Wellington, W.G. (1975) A comparison of the reproductive patterns of apterous and alate virginoparous Acyrthosiphon pisum (Homoptera: Aphididae). Canadian Entomologist, 107, 1161-166.
Mani, M.S. (1962) Introduction to High Altitude Entomology: Insect Life above the Timber-line in the Northwest Himalaya. Methuen, London.
Moore, R.G. & Hanks, L.M. (2004) Aerial dispersal and host plant selection by neonate Thyridopteryx ephemeraeformis (Lepidoptera: Psychidae). Ecological Entomology, 29, 327-335.
Rhainds, M., Leather, S.R. & Sadoff, C. (2008) Polyphagy, flightlessness and reproductive output of females: a case study with bagworms (Lepidoptera: Psychidae). Ecological Entomology, 33, 663-672.
Rhains, M., Davis, D.R. & Price, P.W.(2009) Bionomics of Bagworms (Lepidoptera: Psychidae). Annual Review of Entomology, 54, 209-226.
Roff, D.A. (1990) The evolution of flightlessness in insects. Ecological Monographs, 60, 389-422.
Stjernholm, F., Karlsson, B. & Boggs, C.A. (2005) Age-related changes in thoracic mass: possible reallocation of resources to reproduction in butterflies. Biological Journal of the Linnean Society, 86, 363-380.
Tan, J.Y., Wainhouse, D.W., Day, K.R. & Morgan, G. (2010) Flight ability and reproductive development in newly-emerged pine weevil Hylobius abietis and the potential effects of climate change. Agricultural and Forest Entomology, 12, 427-434.
Tikkanen, O.P., Carr, T.G. & Roininen, H. (1999) Factors influencing the distribution of a generalist spring-feeding moth, Operophtera brumata (Lepidoptera: Geometridae), on host plants. Environmental Entomology, 28, 461-469.
Wagner, D.L. & Liebherr, J.K. (1992) Flightlessness in insects. Trends in Evolution & Ecology, 7, 216-219.
Watt, A.D., Evans, R. & Varley, T. (1992) The egg-laying behaviour of a native insect, the winter moth Operophtera brumata (L.) (Lep., Geometridae), on an introduced tree species, Sitka spruce, Picea sitchensis. Journal of Applied Entomology, 114, 1-4.
Wint, W. (1983) The role of alternative host-plant species in the life of a polyphagous moth, Operophtera brumata (Lepidoptera, Geomtridae). Journal of Animal Ecology, 52, 439-450.
Wollaston, T.V. (1854) Insecta Maderensia, John van Voorst, London.